 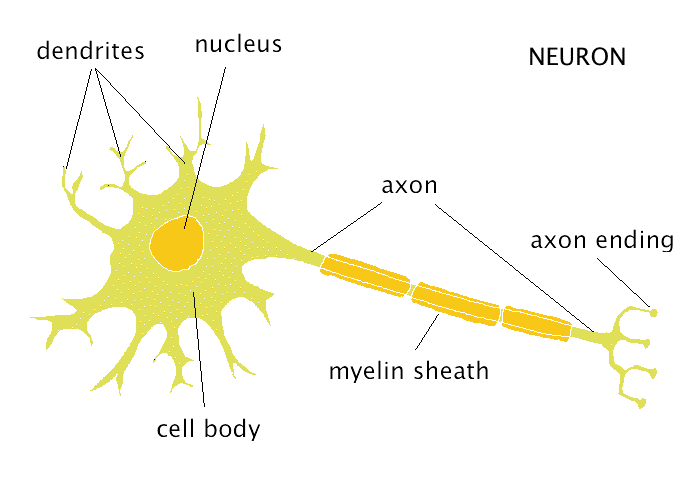
There are three major classes of HDACs, grouped based on their homology to yeast histone deacetylases: class I HDACs bear high homology to the yeast RPD3 protein, class II to Hda1, and class III to Sir2 ( Thiagalingam et al., 2003). For instance, inhibition of HDACs can improve memory in mouse models of neurodegenerative diseases ( Fischer et al., 2007) and can reduce polyglutamine-dependent neurodegeneration in Drosophila ( Steffan et al., 2001). In the nervous system, HDACs have been most intensely studied in neurodegeneration. 
HDACs are best studied for their role in deacetylating lysines on histones, resulting in a more compact chromatin structure correlated with gene repression ( Thiagalingam et al., 2003). We report here a genetic screen for additional factors that regulate PN dendrite wiring specificity, and identify Rpd3 and the transcription factor Prospero (Pros) as additional regulators of PN dendrite wiring specificity. Previous studies have identified several transcription factors that act cell-autonomously to regulate PN dendrite targeting specificity ( Komiyama et al., 2003 Zhu et al., 2006 Komiyama and Luo, 2007 Spletter et al., 2007). Each olfactory projection neuron (PN) targets its dendrites to one of ∼50 glomeruli in the antennal lobe to make synaptic connections with a specific class of olfactory receptor neurons, and sends its axon stereotypically to higher brain centers ( Jefferis et al., 2001 Marin et al., 2002 Wong et al., 2002). The Drosophila olfactory system with its stereotyped organization provides an excellent model to study wiring specificity. Our study suggests a specific function for the general chromatin remodeling factor Rpd3 in regulating dendrite targeting in neurons, largely through the postmitotic action of the Pros transcription factor. Strikingly, overexpression of Pros can suppress Rpd3 −/− phenotypes. Furthermore, disruption of the atypical homeodomain transcription factor Prospero (Pros) yields similar phenotypes, which can be rescued by Pros expression in postmitotic neurons. These phenotypes can be rescued by postmitotic expression of Rpd3 but not HDAC3, the only other class I HDAC in Drosophila. Rpd3 −/− PN dendrites that normally target to a dorsolateral glomerulus mistarget to medial glomeruli in the antennal lobe, and axons exhibit a severe overbranching phenotype. Rpd3 encodes a class I histone deacetylase (HDAC) homologous to mammalian HDAC1 and HDAC2. Using a forward genetic screen, we identified a mutation in Rpd3 that disrupts PN targeting specificity. Each projection neuron (PN) targets its dendrites to a specific glomerulus in the antennal lobe and its axon stereotypically to higher brain centers. 
The Drosophila olfactory system with its stereotyped organization provides an excellent model to study the transcriptional control of dendrite wiring specificity. Compared to the mechanisms of axon guidance, relatively little is known about the transcriptional control of dendrite guidance. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
